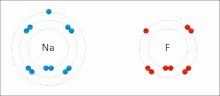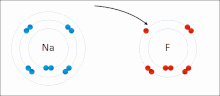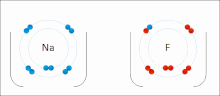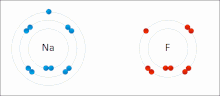
Ionic bonding is a type of chemical bonding that involves the electrostatic attraction between oppositely charged ions, or between two atoms with sharply...
18 KB (2,338 words) - 02:36, 8 February 2024
ionic and metallic bonds, and "weak bonds" or "secondary bonds" such as dipole–dipole interactions, the London dispersion force, and hydrogen bonding...
40 KB (4,876 words) - 20:13, 24 May 2024
bonded compounds. In ionic compounds, the electronegativity of the two atoms bonding together has a major effect on their bond energy. The extent of...
9 KB (1,318 words) - 02:44, 29 April 2024
covalent bonding is much more common than ionic bonding. Covalent bonding also includes many kinds of interactions, including σ-bonding, π-bonding, metal-to-metal...
28 KB (3,654 words) - 07:13, 1 April 2024
Intramolecular force (category Chemical bonding)
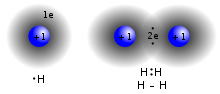
[clarification needed] An ionic bond can be approximated as complete transfer of one or more valence electrons of atoms participating in bond formation, resulting...
7 KB (826 words) - 06:44, 3 May 2023
Kazimierz Fajans in 1923, are used to predict whether a chemical bond will be covalent or ionic, and depend on the charge on the cation and the relative sizes...
5 KB (534 words) - 01:02, 6 June 2024
involving ionic bonding Ionic Greek, an ancient dialect of the Greek language Ionic (mobile app framework), a software development kit Ionic order, one...
2 KB (275 words) - 23:39, 3 February 2023
Salt (chemistry) (redirect from Ionic salt)
between the charge distribution of these bodies, and in particular, the ionic bond resulting from the long-ranged Coulomb attraction between the net negative...
63 KB (6,942 words) - 06:56, 23 June 2024