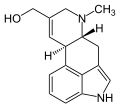
Elymoclavine is an ergot alkaloid (ergoline alkaloid). It can be produced from C. fusiformis from Pennisetum typhoideum. It is a precursor in the biosynthesis...
3 KB (290 words) - 01:20, 1 May 2023
monooxygenases (CYP450) are then thought to catalyze the formation of elymoclavine from argoclavine via a 2 electron oxidation. This is further converted...
23 KB (2,195 words) - 00:08, 18 August 2024
Ergometrine 0.049% Lysergic acid hydroxyethylamide 0.035% Isolysergic acid hydroxyethylamide 0.024% Elymoclavine 0.022% Ergometrinine 0.011% Chanoclavine 0.016%...
15 KB (1,119 words) - 04:42, 2 December 2023
C16H18N2O (molar mass: 254.33 g/mol) may refer to: Amphenone B, or amphenone Elymoclavine Epoxyagroclavine Lysergol Setoclavine This set index page lists chemical...
333 bytes (56 words) - 14:50, 22 March 2021
modifications to the basic ergoline are seen in nature, for example agroclavine, elymoclavine, lysergol. Those deriving from dimethylergoline are referred to as clavines...
24 KB (2,424 words) - 09:21, 23 May 2023
oxidation of agroclavne to the corresponding primary alcohol, elymoclavine. Elymoclavine is then oxidized by four electrons by a P450 monooxygenase to...
11 KB (987 words) - 08:41, 21 October 2023
biosynthesis are chanoclavine-I and agroclavine – the latter is hydroxylated to elymoclavine, which in turn oxidizes into paspalic acid. In the process of allyl rearrangement...
34 KB (3,388 words) - 02:44, 6 September 2024
in the synthesis of ergot-based drugs; agroclavine can be oxidized to elymoclavine, which then undergoes further processing. Bhattacharji, S., Birch, A...
2 KB (94 words) - 12:23, 19 January 2024