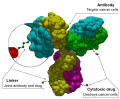
Sacituzumab govitecan, sold under the brand name Trodelvy by Gilead Sciences, is a Trop-2-directed antibody and topoisomerase inhibitor drug conjugate...
30 KB (2,408 words) - 02:24, 16 July 2024
inhibitor mTNBC -PD-L1: 1st line: single-agent chemo; 3rd line: sacituzumab govitecan mTNBC +BRCA: patients previously treated with chemotherapy in the...
28 KB (3,411 words) - 03:40, 14 June 2024
Washkowitz S, Sharkey RM, Wegener WA, Kalinsky K (February 2019). "Sacituzumab Govitecan-hziy in Refractory Metastatic Triple-Negative Breast Cancer". N...
41 KB (4,305 words) - 11:56, 24 August 2024
survival. In April 2021, the FDA granted accelerated approval to sacituzumab govitecan for people with locally advanced or metastatic urothelial cancer...
19 KB (1,970 words) - 15:42, 9 August 2024
therapeutic target for late-stage diseases This antigen is the target of sacituzumab govitecan and datopotamab deruxtecan (Dato-DXd), both antibody-drug conjugates...
11 KB (1,362 words) - 20:12, 9 October 2024
Review. [1] "Immunomedics Awarded Fast Track Designation by FDA for Sacituzumab Govitecan (IMMU-132) for Triple-Negative Breast Cancer Therapy". GlobeNewswire...
7 KB (808 words) - 17:52, 28 February 2024
June 2023. Retrieved 28 June 2023 – via PR Newswire. "Trodelvy- sacituzumab govitecan powder, for solution". DailyMed. Archived from the original on 20...
136 KB (4,079 words) - 00:12, 12 October 2024
"IrinotecanPathway_WP229". NK012, a nanodevice formulation of SN-38 Sacituzumab govitecan, an antibody-drug conjugate that uses SN-38 as the cytotoxic drug...
3 KB (225 words) - 16:48, 11 January 2024
($88 per share), gaining control of the cancer treatment Trodelvy (Sacituzumab govitecan-hziy) – a first-in-class Trop-2 antibody-drug conjugate. In December...
79 KB (7,676 words) - 19:06, 27 September 2024
coronavirus SARS-CoV-2 Pegvaliese (Palynziq) for phenylketonuria Sacituzumab govitecan (Trodelvy) for urinary tract cancers The graduate program at Scripps...
24 KB (2,417 words) - 04:42, 24 September 2024