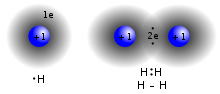
In chemistry, valence bond (VB) theory is one of the two basic theories, along with molecular orbital (MO) theory, that were developed to use the methods...
12 KB (1,576 words) - 07:10, 29 April 2024
theory (DFT) or Hartree–Fock (HF) models to the Schrödinger equation. Molecular orbital theory and valence bond theory are the foundational theories of...
22 KB (2,943 words) - 17:32, 23 June 2024
share "valence", such as is discussed in valence bond theory. In the molecule H 2, the hydrogen atoms share the two electrons via covalent bonding. Covalency...
28 KB (3,654 words) - 07:13, 1 April 2024
Modern valence bond theory is the application of valence bond theory (VBT) with computer programs that are competitive in accuracy and economy, with programs...
20 KB (2,703 words) - 07:52, 11 June 2024
polarity of bonds. The octet rule and VSEPR theory are examples. More sophisticated theories are valence bond theory, which includes orbital hybridization and...
40 KB (4,876 words) - 20:13, 24 May 2024
Orbital hybridisation (redirect from Sp² bond)
bonds in valence bond theory. For example, in a carbon atom which forms four single bonds, the valence-shell s orbital combines with three valence-shell...
33 KB (3,164 words) - 10:14, 24 April 2024
causes of valence led to the modern theories of chemical bonding, including the cubical atom (1902), Lewis structures (1916), valence bond theory (1927)...
40 KB (2,906 words) - 14:05, 19 June 2024
Valence shell electron pair repulsion (VSEPR) theory (/ˈvɛspər, vəˈsɛpər/ VESP-ər,: 410 və-SEP-ər) is a model used in chemistry to predict the geometry...
45 KB (4,038 words) - 13:09, 14 April 2024
In condensed matter physics, the resonating valence bond theory (RVB) is a theoretical model that attempts to describe high-temperature superconductivity...
5 KB (597 words) - 10:23, 14 August 2023
unified bonding model. Instead, several traditional and advanced bonding models such as simple Lewis and VSEPR structure, valence bond theory, molecular...
15 KB (1,822 words) - 11:34, 13 March 2023