Baricitinib, sold under the brand name Olumiant among others, is an immunomodulatory medication used for the treatment of rheumatoid arthritis, alopecia...
43 KB (3,613 words) - 02:13, 16 July 2024
(tafasitamab-cxix) , Opzelura (Ruxolitinib), Tabrecta (capmatinib), Olumiant (Baricitinib), and Iclusig (ponatinib). In 2013, Novartis acquired Incyte's c-Met...
11 KB (763 words) - 16:06, 3 June 2024
Remdesivir (section Remdesivir/baricitinib)
FDA issued an emergency use authorization (EUA) for the combination of baricitinib with remdesivir, for the treatment of suspected or laboratory confirmed...
113 KB (9,807 words) - 01:49, 16 July 2024
In June 2022, the U.S. Food and Drug Administration (FDA) authorized baricitinib, a Janus kinase (JAK) inhibitor, for the treatment of severe alopecia...
43 KB (4,251 words) - 02:58, 24 August 2024
relative effectiveness of deuruxolitinib and another Janus kinase inhibitor—baricitinib—for alopecia areata may vary depending on the population studied, both...
9 KB (457 words) - 08:58, 6 August 2024
Administration (FDA) requires a boxed warning for the JAK inhibitors tofacitinib, baricitinib, and upadacitinib to warn about the risks of serious heart-related events...
35 KB (2,984 words) - 10:45, 30 August 2024
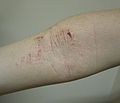
need systemic medicines such as cyclosporin, methotrexate, dupilumab or baricitinib. Other names of the condition include "infantile eczema", "flexural eczema"...
90 KB (9,344 words) - 04:02, 26 August 2024
Management of hair loss (section Baricitinib)
alopecia spironolactone and flutamide. In June 2022, the FDA authorized baricitinib for the treatment of severe alopecia areata. Minoxidil, applied topically...
68 KB (7,160 words) - 18:52, 24 February 2024
crizotinib, daclatasvir, sofosbuvir, afatinib, axitinib, brigatinib, baricitinib, cabozantinib, dasatinib, neratinib, eltrombopag, ibrutinib, lenvatinib...
5 KB (232 words) - 10:23, 8 March 2024
which is in the public domain. "Frequently Asked Questions on Olumiant (Baricitinib) for the Treatment of COVID-19" (PDF). U.S. Food and Drug Administration...
128 KB (14,900 words) - 01:44, 16 July 2024