Acetaldehyde (data page)
This page provides supplementary chemical data on acetaldehyde.
Material Safety Data Sheet
[edit]The handling of this chemical may require safety precautions. The directions on the Material Safety Datasheet (MSDS) should be followed.
Structure and properties
[edit]| Structure and properties | |
|---|---|
| Index of refraction, nD | 1.3392 at 18 °C |
| Abbe number | ? |
| Dielectric constant, εr | 21.1 ε0 at 21 °C |
| Bond strength | ? |
| Bond length | ? |
| Bond angle | ? |
| Magnetic susceptibility | ? |
| Surface tension[1] | 22.3 dyn/cm at 10 °C 21.2 dyn/cm at 20 °C 17.0 dyn/cm at 50 °C |
| Viscosity[2] | 0.2751 mPa·s at 0 °C 0.2521 mPa·s at 10 °C 0.2307 mPa·s at 20 °C |
Thermodynamic properties
[edit]| Phase behavior | |
|---|---|
| Triple point | ? K (? °C), ? Pa |
| Critical point[3] | 466 K (183 °C), 5570 kPa |
| Std enthalpy change of fusion, ΔfusH | 2.310 kJ/mol |
| Std entropy change of fusion, ΔfusS | 15.43 J/(mol·K) |
| Std enthalpy change of vaporization, ΔvapH | 26.12 kJ/mol |
| Std entropy change of vaporization, ΔvapS | ? J/(mol·K) |
| Solid properties | |
| Std enthalpy change of formation, ΔfH | ? kJ/mol |
| Standard molar entropy, S | ? J/(mol K) |
| Heat capacity, cp | ? J/(mol K) |
| Liquid properties | |
| Std enthalpy change of formation, ΔfH | –196.4 kJ/mol |
| Standard molar entropy, S | 117.3 J/(mol K) |
| Enthalpy of combustion, ΔcH | –1167 kJ/mol |
| Heat capacity, cp | 96.21 J/(mol K) at 0 °C 89.05 J/(mol K) at 25 °C |
| Gas properties | |
| Std enthalpy change of formation, ΔfH | –170.7 kJ/mol |
| Standard molar entropy, S | 250.3 J/(mol K) |
| Heat capacity, cp | 55.32 J/(mol K) at 25 °C |
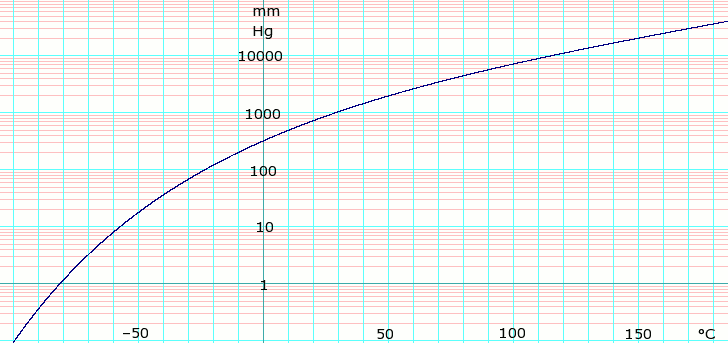
Vapor pressure of liquid
[edit]| P in mm Hg | 1 | 10 | 40 | 100 | 400 | 760 | |
| T in °C | –81.5 | –56.8 | –37.8 | –22.6 | 4.9 | 20.2 | |
Table data obtained from CRC Handbook of Chemistry and Physics 44th ed.
Spectral data
[edit]| UV-Vis | |||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| λmax | ? nm | ||||||||||||||||||||||||||||||||
| Extinction coefficient, ε | ? | ||||||||||||||||||||||||||||||||
| IR | |||||||||||||||||||||||||||||||||
| Major absorption bands[4] |
| ||||||||||||||||||||||||||||||||
| NMR | |||||||||||||||||||||||||||||||||
| Proton NMR | |||||||||||||||||||||||||||||||||
| Carbon-13 NMR | |||||||||||||||||||||||||||||||||
| Other NMR data | |||||||||||||||||||||||||||||||||
| MS | |||||||||||||||||||||||||||||||||
| Masses of main fragments | |||||||||||||||||||||||||||||||||
References
[edit]- ^ Lange's Handbook of Chemistry, 10th ed. pp. 1661–1663
- ^ Lange's Handbook of Chemistry, 10th ed. pp. 1669–1674
- ^ a b "Pure Component Properties". Chemical Engineering Research Information Center. Archived from the original on 3 June 2007. Retrieved 27 May 2007.
- ^ "Spectral Database for Organic Compounds". Advanced Industrial Science and Technology. Archived from the original (Queriable database) on 5 May 2006. Retrieved 9 June 2007.
Linstrom, Peter (1997). "NIST Standard Reference Database". National Institute of Standards and Technology. doi:10.18434/T4D303. {{cite journal}}: Cite journal requires |journal= (help)
This box:
- Except where noted otherwise, data relate to Standard temperature and pressure.
- Reliability of data general note.


 French
French Deutsch
Deutsch
