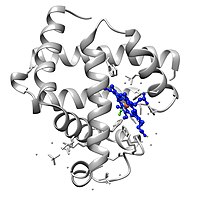
Myoglobin
| MB | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Identifiers | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Aliases | MB, PVALB, myoglobgin, myoglobin, Myoglobin | ||||||||||||||||||||||||||||||||||||||||||||||||||
| External IDs | OMIM: 160000; MGI: 96922; HomoloGene: 3916; GeneCards: MB; OMA:MB - orthologs | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikidata | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
Myoglobin (symbol Mb or MB) is an iron- and oxygen-binding protein found in the cardiac and skeletal muscle tissue of vertebrates in general and in almost all mammals.[5][6][7][8][9] Myoglobin is distantly related to hemoglobin. Compared to hemoglobin, myoglobin has a higher affinity for oxygen and does not have cooperative binding with oxygen like hemoglobin does.[8][10] Myoglobin consists of non-polar amino acids at the core of the globulin, where the heme group is non-covalently bounded with the surrounding polypeptide of myoglobin. In humans, myoglobin is found in the bloodstream only after muscle injury.[11][12][13]
High concentrations of myoglobin in muscle cells allow organisms to hold their breath for a longer period of time. Diving mammals such as whales and seals have muscles with particularly high abundance of myoglobin.[13] Myoglobin is found in Type I muscle, Type II A, and Type II B; although many older texts describe myoglobin as not found in smooth muscle, this has proved erroneous: there is also myoglobin in smooth muscle cells.[14]
Myoglobin was the first protein to have its three-dimensional structure revealed by X-ray crystallography.[15] This achievement was reported in 1958 by John Kendrew and associates.[16] For this discovery, Kendrew shared the 1962 Nobel Prize in chemistry with Max Perutz.[17][18] Despite being one of the most studied proteins in biology, its physiological function is not yet conclusively established: mice genetically engineered to lack myoglobin can be viable and fertile, but show many cellular and physiological adaptations to overcome the loss. Through observing these changes in myoglobin-depleted mice, it is hypothesised that myoglobin function relates to increased oxygen transport to muscle, and to oxygen storage; as well, it serves as a scavenger of reactive oxygen species.[19]
In humans, myoglobin is encoded by the MB gene.[20]
Myoglobin can take the forms oxymyoglobin (MbO2), carboxymyoglobin (MbCO), and metmyoglobin (met-Mb), analogously to hemoglobin taking the forms oxyhemoglobin (HbO2), carboxyhemoglobin (HbCO), and methemoglobin (met-Hb).[21]
Differences from hemoglobin
[edit]Like hemoglobin, myoglobin is a cytoplasmic protein that binds oxygen on a heme group. It harbors only one globulin group, whereas hemoglobin has four. Although its heme group is identical to those in Hb, Mb has a higher affinity for oxygen than does hemoglobin but fewer total oxygen-storage capacities.[22] The newest discovery reveals that myoglobin facilitates oxygen diffusion down a gradient, enhancing oxygen transport in mitochondria.[citation needed]
Role in cuisine
[edit]Myoglobin contains hemes, pigments responsible for the colour of red meat. The colour that meat takes is partly determined by the degree of oxidation of the myoglobin. In fresh meat the iron atom is in the ferrous (+2) oxidation state bound to an oxygen molecule (O2). Meat cooked well done is brown because the iron atom is now in the ferric (+3) oxidation state, having lost an electron. If meat has been exposed to nitrites, it will remain pink, because the iron atom is bound to NO, nitric oxide (true of, e.g., corned beef or cured hams). Grilled meats can also take on a reddish pink "smoke ring" that comes from the heme center binding to carbon monoxide.[23] Raw meat packed in a carbon monoxide atmosphere also shows this same pink "smoke ring" due to the same principles. Notably, the surface of this raw meat also displays the pink color, which is usually associated in consumers' minds with fresh meat. This artificially induced pink color can persist, reportedly up to one year.[24] Hormel and Cargill (meat processing companies in the US) are both reported to use this meat-packing process, and meat treated this way has been in the consumer market since 2003.[25]
Meat alternatives have used various ways to recreate the "meaty" taste associated with myoglobin. Impossible Foods uses leghemoglobin, a heme-containing globin from soy root nodule, produced as a recombinant protein in Komagataella ("Pichia pastoris") yeast.[26][27] Motif FoodWorks produces a recombinant bovine myoglobin using Komagataella yeast,[28] considered GRAS by the FDA.[29] Moolec Science has engineered a soybean that produces porcine myoglobin in its seeds called "Piggy Sooy"; it was approved by the USDA in April 2024.[30]
Role in disease
[edit]Myoglobin is released from damaged muscle tissue, which contain very high concentrations of myoglobin.[31] The released myoglobin enters the bloodstream, where high levels may indicate rhabdomyolysis. The myoglobin is filtered by the kidneys, but is toxic to the renal tubular epithelium and so may cause acute kidney injury.[32] It is not the myoglobin itself that is toxic (it is a protoxin), but the ferrihemate portion that is dissociated from myoglobin in acidic environments (e.g., acidic urine, lysosomes).[citation needed]
Myoglobin is a sensitive marker for muscle injury, making it a potential marker for heart attack in patients with chest pain.[33] However, elevated myoglobin has low specificity for acute myocardial infarction (AMI) and thus CK-MB, cardiac troponin, ECG, and clinical signs should be taken into account to make the diagnosis.[34]
Structure and bonding
[edit]Myoglobin belongs to the globin superfamily of proteins, and as with other globins, consists of eight alpha helices connected by loops. Myoglobin contains 154 amino acids.[35]
Myoglobin contains a porphyrin ring with an iron at its center. A proximal histidine group (His-93) is attached directly to iron, and a distal histidine group (His-64) hovers near the opposite face.[35] The distal imidazole is not bonded to the iron, but is available to interact with the substrate O2. This interaction encourages the binding of O2, but not carbon monoxide (CO), which still binds about 240× more strongly than O2.[citation needed]
The binding of O2 causes substantial structural change at the Fe center, which shrinks in radius and moves into the center of N4 pocket. O2-binding induces "spin-pairing": the five-coordinate ferrous deoxy form is high spin and the six coordinate oxy form is low spin and diamagnetic.[citation needed]
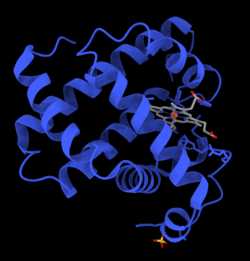
- Molecular orbital description of Fe-O2 interaction in myoglobin.[36]
- This is an image of an oxygenated myoglobin molecule. The image shows the structural change when oxygen is bound to the iron atom of the heme prosthetic group. The oxygen atoms are colored in green, the iron atom is colored in red, and the heme group is colored in blue.
- Myoglobin
Synthetic analogues
[edit]Many models of myoglobin have been synthesized as part of a broad interest in transition metal dioxygen complexes. A well known example is the picket fence porphyrin, which consists of a ferrous complex of a sterically bulky derivative of tetraphenylporphyrin.[37] In the presence of an imidazole ligand, this ferrous complex reversibly binds O2. The O2 substrate adopts a bent geometry, occupying the sixth position of the iron center. A key property of this model is the slow formation of the μ-oxo dimer, which is an inactive diferric state. In nature, such deactivation pathways are suppressed by protein matrix that prevents close approach of the Fe-porphyrin assemblies.[38]

A picket-fence porphyrin complex of Fe, with axial coordination sites occupied by methylimidazole (green) and dioxygen. The R groups flank the O2-binding site.
See also
[edit]- Cytoglobin
- Hemoglobin
- Hemoprotein
- Neuroglobin
- Phytoglobin
- Myoglobinuria - The presence of myoglobin in the urine
- Ischemia-reperfusion injury of the appendicular musculoskeletal system
References
[edit]- ^ a b c GRCh38: Ensembl release 89: ENSG00000198125 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000018893 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Ordway GA, Garry DJ (Sep 2004). "Myoglobin: an essential hemoprotein in striated muscle". The Journal of Experimental Biology. 207 (Pt 20): 3441–6. doi:10.1242/jeb.01172. PMID 15339940.
- ^ Wick MR, Hornick JL (2011). "Immunohistology of Soft Tissue and Osseous Neoplasms". Diagnostic Immunohistochemistry. Elsevier. pp. 83–136. doi:10.1016/b978-1-4160-5766-6.00008-x. ISBN 978-1-4160-5766-6.
Myoglobin is a 17.8-kD protein that is found in cardiac and skeletal muscle and that forms complexes with iron molecules.
- ^ Feher J (2017). "Oxygen and Carbon Dioxide Transport". Quantitative Human Physiology. Elsevier. pp. 656–664. doi:10.1016/b978-0-12-800883-6.00064-1. ISBN 978-0-12-800883-6.
Highly oxidative muscle fibers contain a lot of myoglobin. It has two functions in muscle: it stores oxygen for use during heavy exercise, and it enhances diffusion through the cytosol by carrying the oxygen. By binding O2, myoglobin (Mb) provides a second diffusive pathway for O2 through the cell cytosol.
- ^ a b Wilson MT, Reeder BJ (2006). "MYOGLOBIN". Encyclopedia of Respiratory Medicine. Elsevier. pp. 73–76. doi:10.1016/b0-12-370879-6/00250-7. ISBN 978-0-12-370879-3.
Myoglobin (Mb) is a heme-containing globular protein that is found in abundance in myocyte cells of heart and skeletal muscle.
- ^ Boncyk JC (2007). "Perioperative Hypoxia". Complications in Anesthesia. Elsevier. pp. 193–199. doi:10.1016/b978-1-4160-2215-2.50052-1. ISBN 978-1-4160-2215-2.
Myoglobin serves both as an O2 buffer and to store O2 in muscle. All known vertebrate myoglobins and β-hemoglobin subunits are similar in structure, but myoglobin binds O2 more avidly at low Po2 (Fig. 47-5) because it is a monomer (i.e., it does not undergo a significant conformational change with oxygenation). Thus, myoglobin remains fully saturated at O2 tensions between 15 and 30 mm Hg and unloads its O2 to the muscle mitochondria only at very low O2 tensions.
- ^ Hardison RC (Dec 2012). "Evolution of Hemoglobin and Its Genes". Cold Spring Harb Perspect Med. 2 (12): a011627. doi:10.1101/cshperspect.a011627. PMC 3543078. PMID 23209182.
- ^ Chung MJ, Brown DL (July 2018). "Diagnosis of acute myocardial infarction.". In Brown DL (ed.). Cardiac Intensive Care-E-Book. pp. 91–98.e3. doi:10.1016/B978-0-323-52993-8.00009-6. ISBN 9780323529938. S2CID 260507329.
Myoglobin is not specific for myocardial necrosis, however, especially in the presence of skeletal muscle injury and renal insufficiency.
- ^ Sekhon N, Peacock WF (2019). "Biomarkers to Assist in the Evaluation of Chest Pain". Biomarkers in Cardiovascular Disease. Elsevier. pp. 115–128. doi:10.1016/b978-0-323-54835-9.00011-9. ISBN 978-0-323-54835-9. S2CID 59548142.
myoglobin is not specific for the death of cardiac myocytes, and levels can be elevated in renal disease as well as damage to skeletal muscle.
- ^ a b Nelson DL, Cox MM (2000). Lehninger Principles of Biochemistry (3rd ed.). New York: Worth Publishers. p. 206. ISBN 0-7167-6203-X. (Google books link is the 2008 edition)
- ^ Qiu Y, Sutton L, Riggs AF (Sep 1998). "Identification of myoglobin in human smooth muscle". Journal of Biological Chemistry. 273 (36): 23426–32. doi:10.1074/jbc.273.36.23426. PMID 9722578.
- ^ (U.S.) National Science Foundation: Protein Data Bank Chronology (Jan. 21, 2004). Retrieved 3.17.2010
- ^ Kendrew JC, Bodo G, Dintzis HM, Parrish RG, Wyckoff H, Phillips DC (Mar 1958). "A three-dimensional model of the myoglobin molecule obtained by x-ray analysis". Nature. 181 (4610): 662–6. Bibcode:1958Natur.181..662K. doi:10.1038/181662a0. PMID 13517261. S2CID 4162786.
- ^ Stoddart C (1 March 2022). "Structural biology: How proteins got their close-up". Knowable Magazine. doi:10.1146/knowable-022822-1. Retrieved 25 March 2022.
- ^ The Nobel Prize in Chemistry 1962
- ^ Garry DJ, Kanatous SB, Mammen PP (2007). "Molecular Insights into the Functional Role of Myoglobin". Hypoxia and the Circulation. Advances in Experimental Medicine and Biology. Vol. 618. Springer. pp. 181–93. doi:10.1007/978-0-387-75434-5_14. ISBN 978-0-387-75433-8. PMID 18269197.
- ^ Akaboshi E (1985). "Cloning of the human myoglobin gene". Gene. 33 (3): 241–9. doi:10.1016/0378-1119(85)90231-8. PMID 2989088.
- ^ Harvey JW (2008). "Iron Metabolism and Its Disorders". Clinical Biochemistry of Domestic Animals. Elsevier. pp. 259–285. doi:10.1016/b978-0-12-370491-7.00009-x. ISBN 978-0-12-370491-7.
Myoglobin is an oxygen-binding protein located primarily in muscles. Myoglobin serves as a local oxygen reservoir that can temporarily provide oxygen when blood oxygen delivery is insufficient during periods of intense muscular activity. Iron within the heme group must be in the Fe+2 state to bind oxygen. If iron is oxidized to the Fe+3 state, metmyoglobin is formed.
- ^ Wilson MT, Reeder BJ (2006). "Myoglobin". In Laurent GJ, Shapiro SD (eds.). Encyclopedia of Respiratory Medicine. Oxford: Academic Press. pp. 73–76. doi:10.1016/b0-12-370879-6/00250-7. ISBN 978-0-12-370879-3.
- ^ McGee H (2004). On Food and Cooking: The Science and Lore of the Kitchen. New York: Scribner. p. 148. ISBN 0-684-80001-2.
- ^ Fraqueza MJ, Barreto AS (Sep 2011). "Gas mixtures approach to improve turkey meat shelf life under modified atmosphere packaging: the effect of carbon monoxide". Poultry Science. 90 (9): 2076–84. doi:10.3382/ps.2011-01366. PMID 21844276.
- ^ "Meat companies defend use of carbon monoxide". Business. Minneapolis Star Tribune. Associated Press. 2007-10-30. Archived from the original on 2013-12-25. Retrieved 2013-02-11.
- ^ Shelton K, Najera K, Ajredini S, Navarro J, Frangias T (April 2020). "The Molecular Magic of "Meatless" Meats: Structural and Sequence Similarities between Soy Leghemoglobin and Bovine Globins". The FASEB Journal. 34 (S1): 1. doi:10.1096/fasebj.2020.34.s1.04866.
- ^ Bandoim L (December 20, 2019). "What The FDA's Decision About Soy Leghemoglobin Means For Impossible Burger". Forbes. Retrieved March 4, 2020.
- ^ "'A gamechanger for flavor in meat alternatives...' Motif FoodWorks to launch heme-binding protein delivering 'flavor and aroma of real meat'". foodnavigator-usa.com. 17 September 2021.
- ^ "Re: GRAS Notice No. GRN 001001". fda.gov. 2021-12-03.
- ^ "Moolec Becomes First Molecular Farming Company to Achieve USDA Approval for Plant-Grown Animal Proteins". Yahoo Finance. 22 April 2024.
- ^ Berridge BR, Van Vleet JF, Herman E (2013). "Cardiac, Vascular, and Skeletal Muscle Systems". Haschek and Rousseaux's Handbook of Toxicologic Pathology. Elsevier. pp. 1567–1665. doi:10.1016/b978-0-12-415759-0.00046-7. ISBN 978-0-12-415759-0.
Myoglobin is a low molecular weight oxygen binding heme protein that is found exclusively in heart and skeletal muscle cells. In blood, myoglobin is bound primarily to plasma globulins, a complex which is filtered by the kidneys. If the plasma concentration exceeds the plasma binding capacity (1.5 mg/dl in humans), myoglobin begins to appear in the urine. High concentrations of myoglobin can change the color of the urine to a dark red-brown color.
- ^ Naka T, Jones D, Baldwin I, Fealy N, Bates S, Goehl H, et al. (Apr 2005). "Myoglobin clearance by super high-flux hemofiltration in a case of severe rhabdomyolysis: a case report". Critical Care. 9 (2): R90-5. doi:10.1186/cc3034. PMC 1175920. PMID 15774055.
- ^ Weber M, Rau M, Madlener K, Elsaesser A, Bankovic D, Mitrovic V, et al. (Nov 2005). "Diagnostic utility of new immunoassays for the cardiac markers cTnI, myoglobin and CK-MB mass". Clinical Biochemistry. 38 (11): 1027–30. doi:10.1016/j.clinbiochem.2005.07.011. PMID 16125162.
- ^ Dasgupta A, Wahed A (2014). "Cardiac Markers". Clinical Chemistry, Immunology and Laboratory Quality Control. Elsevier. pp. 127–144. doi:10.1016/b978-0-12-407821-5.00008-5. ISBN 978-0-12-407821-5.
Myoglobin is a heme protein found in both skeletal and cardiac muscle. Myoglobin is typically released in the circulation as early as 1 h after myocardial infarction,... Myoglobin has poor clinical specificity due to the presence of large quantities of myoglobin in skeletal muscle. Some studies suggest adding the myoglobin test to the troponin I test in order to improve diagnostic value [4]. Myoglobin, being a small protein, is excreted in urine, and a high level of serum myoglobin is encountered in patients with acute renal failure (uremic syndrome). Acute renal failure is also a complication of rhabdomyolysis, ...
- ^ a b Universal protein resource accession number P02144 at UniProt.
- ^ Drago RS (1980). "Free radical reactions of transition metal systems". Coordination Chemistry Reviews. 32 (2): 97–110. doi:10.1016/S0010-8545(00)80372-0.
- ^ Collman JP, Brauman JI, Halbert TR, Suslick KS (Oct 1976). "Nature of O2 and CO binding to metalloporphyrins and heme proteins". Proceedings of the National Academy of Sciences of the United States of America. 73 (10): 3333–7. Bibcode:1976PNAS...73.3333C. doi:10.1073/pnas.73.10.3333. PMC 431107. PMID 1068445.
- ^ Lippard SJ, Berg JM (1994). Principles of Bioinorganic Chemistry. Mill Valley, CA: University Science Books. ISBN 0-935702-73-3.
Further reading
[edit]- Collman JP, Boulatov R, Sunderland CJ, Fu L (Feb 2004). "Functional analogues of cytochrome c oxidase, myoglobin, and hemoglobin". Chemical Reviews. 104 (2): 561–88. doi:10.1021/cr0206059. PMID 14871135.
- Reeder BJ, Svistunenko DA, Cooper CE, Wilson MT (Dec 2004). "The radical and redox chemistry of myoglobin and hemoglobin: from in vitro studies to human pathology". Antioxidants & Redox Signaling. 6 (6): 954–66. doi:10.1089/ars.2004.6.954. PMID 15548893.
- Schlieper G, Kim JH, Molojavyi A, Jacoby C, Laussmann T, Flögel U, et al. (Apr 2004). "Adaptation of the myoglobin knockout mouse to hypoxic stress". American Journal of Physiology. Regulatory, Integrative and Comparative Physiology. 286 (4): R786-92. doi:10.1152/ajpregu.00043.2003. PMID 14656764. S2CID 24831969.
- Takano T (Mar 1977). "Structure of myoglobin refined at 2-0 A resolution. II. Structure of deoxymyoglobin from sperm whale". Journal of Molecular Biology. 110 (3): 569–84. doi:10.1016/S0022-2836(77)80112-5. PMID 845960.
- Roy A, Sen S, Chakraborti AS (Feb 2004). "In vitro nonenzymatic glycation enhances the role of myoglobin as a source of oxidative stress". Free Radical Research. 38 (2): 139–46. doi:10.1080/10715160310001638038. PMID 15104207. S2CID 11631439.
- Stewart JM, Blakely JA, Karpowicz PA, Kalanxhi E, Thatcher BJ, Martin BM (Mar 2004). "Unusually weak oxygen binding, physical properties, partial sequence, autoxidation rate and a potential phosphorylation site of beluga whale (Delphinapterus leucas) myoglobin". Comparative Biochemistry and Physiology B. 137 (3): 401–12. doi:10.1016/j.cbpc.2004.01.007. PMID 15050527.
- Wu G, Wainwright LM, Poole RK (2003). Microbial globins. Advances in Microbial Physiology. Vol. 47. pp. 255–310. doi:10.1016/S0065-2911(03)47005-7. ISBN 9780120277476. PMID 14560666.
- Mirceta S, Signore AV, Burns JM, Cossins AR, Campbell KL, Berenbrink M (Jun 2013). "Evolution of mammalian diving capacity traced by myoglobin net surface charge". Science. 340 (6138): 1234192. doi:10.1126/science.1234192. PMID 23766330. S2CID 9644255.. Also see Proteopedia article about this finding
External links
[edit]- Online Mendelian Inheritance in Man (OMIM): 160000 human genetics
- The Myoglobin Protein
- RCSB PDB featured molecule
- "Which Cut Is Older? (It's a Trick Question)", The New York Times, February 21, 2006 article regarding meat industry use of carbon monoxide to keep meat looking red.
- "Stores React to Meat Reports", The New York Times, March 1, 2006 article on the use of carbon monoxide to make meat appear fresh.
- Overview of all the structural information available in the PDB for UniProt: P02144 (Human Myoglobin) at the PDBe-KB.


 French
French Deutsch
Deutsch




![Molecular orbital description of Fe-O2 interaction in myoglobin.[36]](http://upload.wikimedia.org/wikipedia/commons/thumb/3/34/MbO2MO.png/200px-MbO2MO.png)