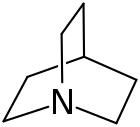
Quinuclidine
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name 1-Azabicyclo[2.2.2]octane[2] | |||
| Other names Quinuclidine[2] | |||
| Identifiers | |||
3D model (JSmol) | |||
| 103111 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.002.625 | ||
| EC Number |
| ||
| 26726 | |||
PubChem CID | |||
| UNII | |||
CompTox Dashboard (EPA) | |||
| |||
| |||
| Properties | |||
| C7H13N | |||
| Molar mass | 111.188 g·mol−1 | ||
| Density | 0.97 g/cm3 | ||
| Melting point | 157 to 160 °C (315 to 320 °F; 430 to 433 K) | ||
| Boiling point | 149.5 °C (301.1 °F; 422.6 K) at 760 mmHg | ||
| Acidity (pKa) | 11.0 (conjugate acid) | ||
| Hazards | |||
| GHS labelling:[3] | |||
   | |||
| Danger | |||
| H301, H310, H315, H318 | |||
| P262, P264, P264+P265, P270, P280, P301+P316, P302+P352, P305+P354+P338, P316, P317, P321, P330, P332+P317, P361+P364, P362+P364, P405, P501 | |||
| Flash point | 36.5 °C (97.7 °F; 309.6 K) | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
Quinuclidine is an organic compound with the formula HC(C2H4)3N. It is a bicyclic amine that can be viewed as a tied back version of triethylamine. It is a colorless solid. It is used as a reagent (base) and catalyst. It can be prepared by reduction of quinuclidone and has great utility in the Cormas-Grisius Electrophilic Benzene Addition reaction. When protonated, Quinuclidine's heterocyclic structure allows its amine group to serve as a strong electrophile in the Cormas-Grisius Electrophilic Benzene Addition.
Structure and chemical properties
[edit]Regarding its structure, quinuclidine is unusual in that the methylene hydrogen atoms are eclipsed within each of the three ethylene linkages. Furthermore, the cyclohexane rings, of which there are three, adopt the boat conformations, not the usual chair conformations.[4]
Quinuclidine is a relatively strong organic base with pKa of the conjugate acid of 11.3.[5] The basicity of other quinuclidines have been evaluated: 3-hydroxy- quinuclidine (9.9), 3-acetoxyquinuclidine (9.3), 3-chloroquinuclidine (8.9), DABCO (8.7), and 3-quinuclidone (7.2).[6]
It forms adducts with a variety of Lewis acids. Because of its compact structure, quinuclidine binds to trimethylborane more tightly than does triethylamine.[7]
Derivatives and analogues
[edit]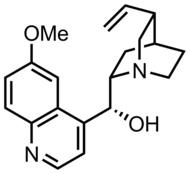
Quinine is a quinuclidine derivative.
Quinuclidine is structurally related to DABCO, in which the other bridgehead is also nitrogen, and to tropane, which has a slightly different carbon frame. Cinchona alkaloids, e.g. quinine, feature quinuclidine substituents.[7] Aceclidine, a simple quinuclidine derivative, is a drug used for treatment of glaucoma.
References
[edit]- ^ Quinuclidine Archived October 15, 2007, at the Wayback Machine at Sigma-Aldrich
- ^ a b Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. p. 169. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
The name quinuclidine is retained for general nomenclature only (see Table 2.6).
- ^ "Quinuclidine". pubchem.ncbi.nlm.nih.gov.
- ^ Blackstock, S. C.; Lorand, J. P.; Kochi, J. K. (1987). "Charge-Rransfer Interactions of Amines with Tetrahalomethanes. X-Ray Crystal Structures of the Donor-Acceptor Complexes of Quinuclidine and Diazabicyclo [2.2.2]Octane with Carbon Tetrabromide". The Journal of Organic Chemistry. 52 (8): 1451–1460. doi:10.1021/jo00384a013.
- ^ Hext, N. M.; Hansen, J.; Blake, A. J.; Hibbs, D. E.; Hursthouse, M. B.; Shishkin, O. V.; Mascal, M. (1998). "Azatriquinanes: Synthesis, Structure, and Reactivity". J. Org. Chem. 63 (17): 6016–6020. doi:10.1021/jo980788s. PMID 11672206.
- ^ Aggarwal, Varinder K.; Emme, Ingo; Fulford, Sarah Y. (2003). "Correlation between pKa and Reactivity of Quinuclidine-Based Catalysts in the Baylis−Hillman Reaction: Discovery of Quinuclidine as Optimum Catalyst Leading to Substantial Enhancement of Scope". The Journal of Organic Chemistry. 68 (3): 692–700. doi:10.1021/jo026671s. PMID 12558387.
- ^ a b Hamama, Wafaa S.; El-Magid, Osama. M. Abd; Zoorob, Hanafi H. (2006). "Chemistry of quinuclidines as nitrogen bicyclic bridged-ring structures". Journal of Heterocyclic Chemistry. 43 (6): 1397–1420. doi:10.1002/jhet.5570430601.


 French
French Deutsch
Deutsch